Qualification and validation of laboratory autoclaves
Is your autoclave really sterilizing?
The necessity of qualifying an autoclave and validating the sterilization process arises from various laws and regulations. Although the terms qualification and validation are usually mentioned in  one sentence or even used synonymously, different statements and approaches are hidden behind them.
one sentence or even used synonymously, different statements and approaches are hidden behind them.
The necessity to qualify an autoclave and to validate the sterilization process comes from, among other things, the Medicines Act, the Medical Devices Act, the Chemicals Act, and the Ordinance on the Protection of Biological Substances and Genetic Engineering. These laws and regulations sometimes refer to directives, rulings, or decisions of the commission, guidelines, and standards. By referring legally binding EU regulations to legally non-binding standards or guidelines such as the EU-GxP guidelines for
- Good Manufacturing Practice (GMP)
- Good Clinical Practice (GCP)
- Good Laboratory Practice (GLP)
they are given a law-like character and their compliance is checked and periodically controlled by government agencies. The aim is to ensure and guarantee a high and above all uniform quality standard. How the individual regulations are differentiated from each other is shown in Figure 1.![]()
According to the EU-GxP guidelines, systems and processes that are directly involved in the manufacturing process for product groups such as pharmaceuticals, active ingredients, cosmetics, food, animal feed, and medical devices and thus have an influence on product quality must be qualified and validated. The requirements of the Guide to Good Laboratory Practice are addressed at laboratories conducting non-clinical studies on test objects such as pharmaceuticals, pesticides, biocides, cosmetics, veterinary medicinal products, food additives, feed additives, and industrial chemicals. Systems and processes used to inactivate human sample materials (body fluids, tissues, cell cultures, etc.), microorganisms, cell cultures, endoparasites – including their genetically modified forms – also fall under this category.
A requirement of the principles of good laboratory practice is that equipment, including validated computer-aided systems used to collect, record, and reproduce data and to control the environmental conditions relevant to the test, must be of adequate design and performance. If your autoclave is directly involved in studies on the above-mentioned product groups, its suitability in terms of design and performance must be proven. In detail, the supervisory authorities will define the requirements for qualification and validation implementation and documentation in more detail by specifying standards, guidelines, etc. or they may follow standards because the standardisation also moves within certain limits.
Qualification and Validation
With such importance placed on the autoclave how can the user be certain that it has achieved the required sterilization?
As an autoclave operator involved in your manufacturing process – or as a laboratory operator – you are responsible for your manufacturing process or for the results and conclusions of your study. Qualification and validation is often carried out with the help of an appropriately qualified service provider. Since only you can define the product-specific requirements for your process, close cooperation between your specialist departments, the qualifier/validator, and the autoclave manufacturer is necessary.
Although the terms qualification and validation are usually mentioned in one sentence or even used synonymously, different statements and approaches are hidden behind them. The common denominator of both concepts is that documents conforming to the guidelines, which contain the definition of the individual tests and the documentation of the test results, must be prepared. In many cases, the written qualification/validation plan also includes protocols that are used to document the test results during implementation.
Qualification: In the context of a system qualification, proof must be provided that a system with the technology used is suitable for the intended task, works perfectly, and delivers results that meet the requirements. The main focus here is therefore on the system (the autoclaves) and its correct function.
Validation: The objective of process validation is to prove that the process is effective and can deliver a product (result) that is reproducible and meets the required quality require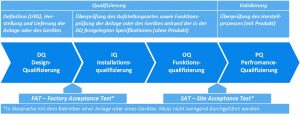 ments. The main focus is therefore on the process itself and proof of compliance with the acceptance criteria that is to be defined by the system operator.
ments. The main focus is therefore on the process itself and proof of compliance with the acceptance criteria that is to be defined by the system operator.
The process qualification is often a process that takes place within a system, which explains the linking of terms. The qualification and validation process is illustrated in Figure 2.
DQ – Design Qualification
The first qualification stage of an autoclave starts as soon as you formulate and document your requirements for the device to be purchased. This means you have already created your User Requirement Specification (URS); i.e. the requirements that are made of a device and which must be fulfilled. For many manufacturers, autoclaves can be equipped with options that influence process accuracy or speed, for example. If certain options depending on the processes to be carried out are not taken into account in the URS, and if they are not upgraded according to the device, this can make successful qualification and validation considerably more difficult or even completely impossible later on. Even at this early stage, future operators should therefore specify exactly what the autoclave is to be used for, what requirements are to be placed on the processes, and which standards and guidelines must be observed.
Depending on the complexity of the system or the autoclave, the scope of the URS will vary greatly. Autoclave suppliers will submit an offer on this basis and, in coordination between the operator and manufacturer, specifications will be drawn up. Checking for conformity is an elementary component of the first qualification level – the Design Qualification (DQ). After a successful DQ, the device is manufactured by the manufacturer according to the agreed specifications. At the same time (either internally, with the manufacturer, or with a separate service provider), preparing the documents required for the subsequent steps IQ, OQ, and PQ (validation plan) can now be started. These documents form the basis for implementing the IQ, OQ, and PQ and specify the inspections and tests to be carried out as well as their parameters and acceptance criteria. Detailed documentation is required to prove that all checks and tests have been successfully completed.
IQ – Installation Qualification
After the design qualification has been successfully completed and the device has been successfully delivered, the next qualification stage of the autoclave can begin on site – the installation qualification (IQ). The suitability of the installation site, the physical characteristics of the supplied autoclave, as well as the completeness of the documentation belonging to the device are now checked and documented in the qualification documents. Documentation generally includes the operating instructions, maintenance, cleaning, and calibration instructions as well as material certificates and data sheets for individual installed components.
If this qualification level has also been successfully completed, or if any deviations that may have been detected have been evaluated on the basis of a risk analysis and appropriate measures have been initiated to eliminate them, the functional qualification (OQ) of the system can be started.
OQ – Operational Qualification
Proof is now provided that the devices, installations, utilities, and systems meet the requirements and specifications of the operator, which were specified within the scope of the design qualification. Tests are carried out to prove that the device is working according to plan. This includes calibration as well as simulating fault, interference, and shutdown conditions of the device. This procedure serves as documented proof that defects which may be safety-relevant or quality-influencing for the product are correctly detected by the autoclave.
Implementing sterilization runs of the autoclave without introduced product is also part of the scope of the functional qualification. Temperature sensors and a pressure sensor are inserted into the autoclave for this, which use a data logger to measure pressure and temperature independently of the autoclave controller. The temperature and pressure values determined in this way can thus be checked against the values determined via the autoclave control. In addition, bioindicators based on Bacillus stearothermophilus are introduced into the autoclaves to prove the biological effectiveness of the sterilization process. The aim is to provide documented proof that a system or device is suitable for its intended use without a product.
PQ- Performance Qualification
Once the autoclave has been proven to function without a product, the performance of the device is checked according to the specifications of the validation plan defined in advance, with the aim of proving that the specified process requirements are adequately met under real conditions (with a product). It should be noted that the temperature distribution depends on the product to be sterilised and therefore a separate PQ test must be performed for each loading configuration. Loading patterns are defined for this which should be representative of the loads to be expected in everyday operation. This is often done according to the following scheme:
- Minimum loading,
- Medium loading,
- Worst case loading.
Worst case loading absolutely must be checked with regard to the loading quantity, for example. Other parameters may need to be considered if they could influence the result of the sterilization process.
As with the OQ, the sterilization process is checked by temperature and pressure sensors which record the corresponding data independently of the autoclave control by means of a data logger. In addition, the biological effectiveness of the sterilization process is also demonstrated by reference indicators based on Bacillus sterothermophilus. In the PQ, unlike in the OQ, the temperature sensors as well as the bioindicators are placed in the product to be tested (loading pattern) in order to provide proof of the sterilizing effectiveness in the product. It is important that the temperature sensors as well as the bioindicators are placed in the points of the product where it is expected that a sterilizing effect is most difficult to achieve. The aim is documented proof that a process can reproducibly deliver the expected result taking the product into account.
When performing the OQ and PQ, the number of temperature measurement points within the working chamber should comply with the recommendations of the relevant standards such as EN 556 or the GxP guidelines, as any deviation from these will probably lead to queries during an inspection. 12 temperature measuring points should therefore be provided per 1 m3 of sterilization chamber. A bioindicator based on the Bacillus stearothermophilus is placed at each temperature measuring point.
Sterility Assurance Level
The EN 556 standard also provides information on when a product can be described as sterile or when a sterilization process is considered reproducible, i.e. always delivers the same result (a sterile product). What is called the Sterility Assurance Level (SAL) is defined here. The SAL assumes a theoretical population of 100 000 (105) microorganisms (Colony Forming Units – CFU), for example.
Bacillus stearothermophilus is used as a reference indicator (bioindicator) for testing steam sterilization processes. The SAL now demands that, depending on the product to be sterilised, the greatest possible probability must be achieved that all microorganisms will actually be killed and thus all products are actually sterile. This greatest possible probability is considered proven if an SAL value of 10-6 is reached. This means that the probability that a product is still non-sterile after the sterilization process is 1: 1 000 000.
Re-Validation
If all qualification steps and the process validation(s) have been successfully completed, the operator must specify an interval for re-validation. It is a good idea to coordinate with the autoclave manufacturer at this point, as it makes sense to combine the re-validation with the necessary recurring calibration of the instrument sensors.
Caution is required when changing process parameters. This includes, for example
- Changing device parameters (e.g. changing sterilisation temperature and/or time);
- Changes in the loading configuration or changes in the composition of the product to be sterilised,
- Changes in packaging for packaged goods,
- Repairs to equipment and the associated replacement of components,
- Device software updates.
If the above mentioned changes occur, their influence on the validated overall process must be examined. If it is to be expected that the change could have an influence on the overall process, the influence must be determined, evaluated, and documented as part of re-validation. This may result in necessary adjustments to the overall process so that it can continue to comply with the parameters defined before the change.
Since changes can also occur gradually, i.e. barely or even not at all noticeably, re-validation should always be carried out at fixed intervals. An interval of one year has been proven here. For particularly critical processes, however, the interval for re-validation can also be defined at shorter intervals.
This article was published in the LABO Magazine 05/2018.
More information about Evidencia GmbH can be found here.